 全部商品分类
全部商品分类

 下载产品说明书
下载产品说明书 下载SDS
下载SDS 用小程序,查商品更便捷
用小程序,查商品更便捷


 收藏
收藏
 对比
对比 咨询
咨询Scientific Data
 View Larger
View LargerCarrier Free
CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
3344-NG
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage: | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
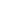
Recombinant Human Noggin Fc Chimera Protein, CF Summary
Product Specifications
| Human Noggin (Gln28-Cys232) Accession # Q13253 | IEGRMD | Human IgG1 (Pro100-Lys330) |
| N-terminus | C-terminus | |
Analysis

Background: Noggin
Noggin is a secreted homodimeric glycoprotein that is an antagonist of bone morphogenetic proteins (BMPs) (1, 2). Human Noggin cDNA encodes a 232 amino acid (aa) precursor protein; cleavage of a 19 aa signal peptide generates the 213 aa mature protein which contains an N-terminal acidic region, a central basic heparin‑binding segment and a C-terminal cysteine-knot structure (2). Secreted Noggin probably remains close to the cell surface due to its binding of heparin‑containing proteoglycans (3). Noggin is very highly conserved among vertebrates, such that mature human Noggin shares 99%, 99%, 98%, 97% and 89% aa sequence identity with mouse, rat, bovine, equine and chicken Noggin, respectively. Noggin binds some BMPs such as BMP-4 with high affinity and others such as BMP-7 with lower affinity, antagonizing BMP bioactivities by blocking epitopes on BMPs that are needed for binding to both type I and type II receptors (2, 4).
During embryogenesis, Noggin antagonizes specific BMPs at defined times during neural tube, somite and cardiomyocyte growth and patterning (5-7). During skeletal development, Noggin prevents chondrocyte hyperplasia, thus allowing proper formation of joints (4). Mutations within the cysteine-knot region of human Noggin are linked to multiple types of skeletal dysplasias that result in apical joint fusions (8). Noggin is expressed in defined areas of the adult central nervous system and peripheral tissues such as lung, skeletal muscle and skin (1). During culture of human embryonic stem cells (hESC) without feeder layers or conditioned medium, but with addition of FGF basic, addition of Noggin to antagonize BMP activity allows hESC to maintain their undifferentiated, pluripotent state (9, 10). In differentiation protocols, Noggin has been used to create neural crest stem cells from induced pluripotent stem cells (11).
Because of its importance in the development of tissues, regenerative medicine utilizes Noggin to generate cells for intestinal tissues or organoids in vitro (12). Noggin is also an important factor for stimulating bone development and has neuroprotective effects in early stages of spinal cord injury (13, 14). Expression of Noggin can help contain or reduce metastatic lesions by limiting BMP signaling, making it a therapeutic option for cancer treatment (15). Noggin has been used to create bladder cancer organoids that can serve as a tissue model in preclinical testing of chimeric antigen receptor (CAR)-T-cell immunotherapy (16).
- Valenzuela, D.M. et al. (1995) J. Neurosci. 15:6077.
- Groppe, J. et al. (2002) Nature 420:636.
- Paine-Saunders, S et al. (2002) J. Biol. Chem. 277:2089.
- Brunet, L. J. et al. (1998) Science 280:1455.
- McMahon, J. A. et al. (1998) Genes Dev. 12:1438.
- Itsykson, P. et al. (2005) Mol. Cell. Neurosci. 30:24.
- Yuasa, S. et al. (2005) Nat. Biotechnol. 23:607.
- Gong, Y. et al. (1999) Nat. Genet. 21:302.
- Xu, R.-H. et al. (2005) Nat. Methods 2:185.
- Wang, G. et al. (2005) Biochem. Biophys. Res. Commun. 330:934.
- Abe, R. et al. (2021) J. Rural. Med. 16:143.
- Kim, S. et al. (2022) Nat. Commu. 13:1692.
- Malijauskaite, S. et al. (2021) Cytokines Growth Factor Rev. 60:76.
- Al-Sammarraie, N. et al. (2022) Neural Regen Res. 18:492.
- Davis, H. et al. (2016) Cytokines Growth Factor Rev. 27:81.
- Yu, L. et al. (2021) Clin. Transl. Immunology 10:e1248.







